The different pharmaceutical formulations with phospholipids excipients
There are many pharmaceutical formulations that contain phospholipids as excipients. You can learn more about these formulations, namely liposomes, emulsions, (mixed) micelles, and lipid nanoparticles in the following article.
Which formulations can be made with phospholipids?
Phospholipids, both natural and synthetic, serve broadly as excipients in pharmaceutical technology, functioning as wetting agents, emulsifiers, and builders of various lipid mesophases such as closed lipid vesicles (liposomes), micelles, inverted micelles.1) These functional properties apply to many types of pharmaceutical formulations, including suspensions, various types of emulsions, solid dispersions, lipid nanoparticles (LNPs), and drug/phospholipid complexes.2)3) Consequently, such formulation types can appear in any dosage form, such as tablets, capsules, injections, and creams.
Given their physiological role, phospholipids are well tolerated and suitable for multiple administration routes, namely parenteral, oral, topical (dermal) and inhalation.
Which phospholipid for which application?
When natural phospholipids derived from hen egg yolk or soybean are selected, one must pay attention to the minimal quality in phosphatidylcholine (PC) content. The required quality depends on the administration route. For oral and dermal administration, natural phospholipids with at least 45% PC can be used, whereas parenteral applications typically require phospholipid fractions with at least 70% PC.
For specific high-tech injectable parenteral products, more expensive, synthetic, and chemically well-defined phospholipids of high purity may offer the best performance. On the other hand, for topical and oral administration - as well as other parental applications - cost-effective natural phospholipids are preferable and approved in marketed drug products.
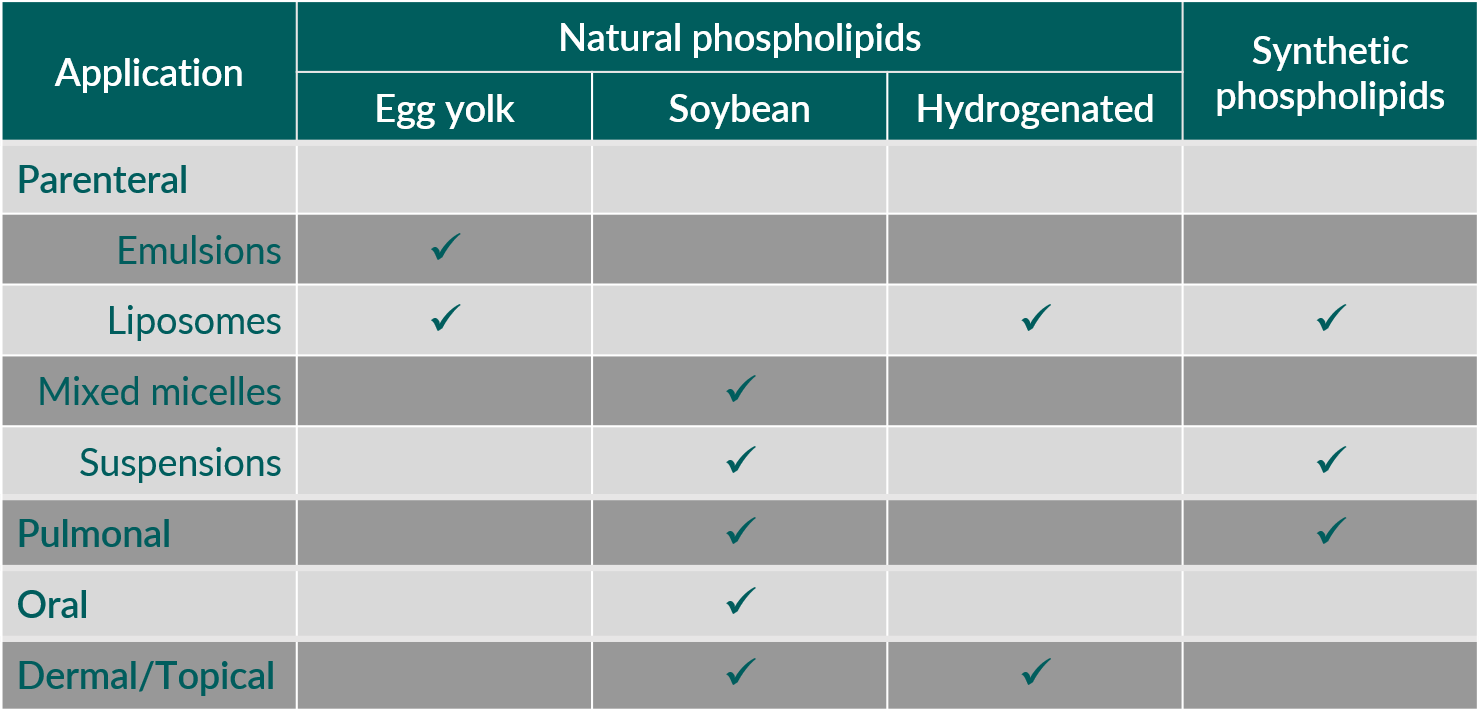
Table 1. Phospholipids in pharmaceutical applications: Which phospholipid is used?
The categories of pharmaceutical formulations
To study the prevalence of natural and synthetic phospholipids, one should focus on products used for parenteral administration.1) These mainly include liposomes, oil-in-water (o/w) emulsions, mixed micelles for intravenous (i.v.) use and slow release, vaccine vehicles, and drug suspensions for intramuscular (i.m.) and subcutaneous (s.c.) administration.
Liposomal products for i.v. administration incorporate both synthetic and natural phospholipids. Furthermore, liposomes can act as vehicles for slow release after local parenteral administration, for example at the surgical site, epidural, or intrathecal.
Typically, o/w emulsions for parenteral nutrition and as carriers for oil-soluble drug substances are prepared with egg phospholipids.4)
In mixed micellar formulations containing phospholipids and various cholate salts, only soybean phospholipids are used. Here, phospholipids function as solubilizer for poorly water-soluble substances or act as active principles (e.g., soybean PC), with the presence of polyunsaturated fatty acids (PUFAs) supporting the treatment of liver disorders.5)
Regarding products with phospholipids for pulmonary administration, both natural and synthetic phospholipids are applied. Most of these products treat respiratory distress syndrome in infants6) or bacterial lung infections.
Regulatory and safety aspects
Natural phospholipids are generally well-known to regulatory authorities. Their long-standing record as excipients demonstrates very high tolerability and biocompatibility. The World Health Organization (WHO) places no limit on the oral lecithin intake, and it also specifies no limit for the value of acceptable daily intake (ADI) for lecithin as a food additive.
Pediatric oral use of phospholipids (soybean) is generally allowed, with appropriate precautions for soybean allergy and with respect to maximum permitted levels, e.g., in dietary foods for infants. Alternatively, sunflower phospholipids, with no allergy warnings, can be used. After parenteral administration, both egg and soybean phospholipids—unsaturated and saturated variations—are very well tolerated.
The European Commission classifies lecithin as a food additive (E322) “generally permitted for use in foodstuffs”. Furthermore, Europe does not set an ADI value for lecithin; it may be used in food quantum satis. The US Food and Drug Administration (FDA) assigns lecithin the "generally recognized as safe" (GRAS) affirmation, including enzyme-modified lecithin.
Pharmacopoeias yet only list a limited number of synthetic phospholipids. However, similar to hydrogenated natural phospholipids, they appear in the FDA Center for Drug Evaluation's Inactive Ingredient Guide and in the relevant reference documents of both FDA and the European Medicines Agency (EMA). Consequently, the quality and safety of synthetic phospholipids must be addressed in the application documentation of the drug product.1)
You want to know more?
Please use the following links:
What phospholipid types are there?
What phospholipid aggregates are formed?
What is the difference between natural and synthetic phospholipids?
What is the natural occurrence and phospholipid benefit?
The use of natural and synthetic phospholipids as pharmaceutical excipients
Eur. J. Lipid Sci. Technol. 116, 1088-1107
| PubMed |
Role of phospholipids in the oral and parenteral delivery of poorly water soluble drugs
J. Drug Delivery Sci. Technol. 21, 5-16
| Elsevier |
Increasing drug solubility by means of bile salt-phosphatidylcholine-based mixed micelles
Eur. J. Pharm. Biopharm. 46, 361-367
| PubMed |
Bovine surfactant therapy for patients with acute respiratory distress syndrome
Am. J. Respir. Crit. Care Med. 155, 1309-1315
| PubMed |

